Klinische ondersteuning & informatiebronnen voor de behandeling van COVID-19
Krijg toegang tot ondersteuning voor producten, waaronder informatie over reiniging en desinfectie, videotutorials, en meer.

Producten en diensten voor de behandeling van COVID-19
Ontdek een uitgebreid portfolio van product- en dienstenoplossingen voor de behandeling van COVID-19-patiënten.
Informatiebronnen voor professionals in de gezondheidszorg

Klinische ondersteuning & informatiebronnen voor de behandeling van COVID-19
Krijg toegang tot ondersteuning voor producten, waaronder informatie over reiniging en desinfectie, videotutorials, en meer.

Producten en diensten voor de behandeling van COVID-19
Ontdek een uitgebreid portfolio van product- en dienstenoplossingen voor de behandeling van COVID-19-patiënten.
Veelgebruikte klinische ondersteuning en middelen
Documenten
Documenten
Veelgestelde vragen over COVID-19-zorg
- Welke filters moeten worden gebruikt voor onze niet-invasieve en invasieve beademingsapparaten?
-
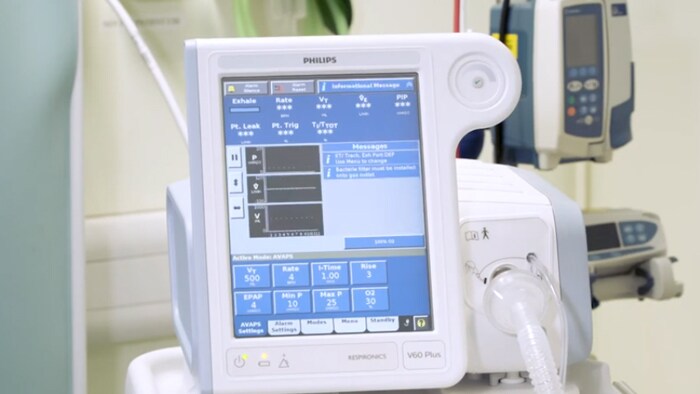
Het is belangrijk om op te merken dat het V60 / V60 Plus-beademingsapparaat is ontworpen met drie filterlagen: het luchtinlaatfilter, het mainflow-bacteriefilter en een filter op de uitademingspoort van het patiëntcircuit. We adviseren klanten om de instructies in de gebruikershandleiding van het V60 / V60 Plus-beademingsapparaat te volgen om lucht te filteren voor levering aan de patiënt met het luchtinlaatfilter, om altijd een mainflow-bacteriefilter op de gasuitlaatpoort van de patiënt te gebruiken, en om een filter op de uitademingspoort van het patiëntcircuit te gebruiken.
- Kan de V60 worden gebruikt voor invasieve beademing?
-
De V60 heeft zijn waarde bewezen in zowel invasieve als niet-invasieve beademing. De Philips Respironics V60 Ventilator is een microprocessorgestuurd beademingssysteem met bilevel-positieve luchtwegdruk (BiPAP). Dit apparaat levert niet-invasieve positieve drukbeademing en invasieve beademing voor spontaan ademende volwassen- en pediatrische patiënten.
- Biedt de uitademingsfiltratie 100% bescherming tegen uitgeademd gas van patiënten?
-
Hoewel het uitgeademde gas dat door de uitademingspoort stroomt effectief wordt gefilterd (> 99,99% efficiëntie), zal bij de meeste patiëntinterfaces een bepaalde hoeveelheid gas lekken als gevolg van een onbedoelde opening waar het kussen in contact is met de huid.
- Hoe vaak moet het bacteriële filter worden vervangen?
-
De frequentie en timing van filtervervanging is afhankelijk van de patiënt en het gebruik. Het wordt aanbevolen om het filter te vervangen tussen patiënten en met regelmatige tussenpozen (of zoals wordt aangegeven door de fabrikant).
- Kan ik ziekenhuisdoekjes met bleekmiddel gebruiken voor Philips/Respironics-ziekenhuismaskers voor gebruik bij meerdere patiënten?
-
Leest u de volledige gebruiksaanwijzing, waarin het volgende staat: Gebruik geen bleekmiddel, alcohol, schoonmaakmiddelen die bleekmiddel of alcohol bevatten, of schoonmaakmiddelen die conditioners of vochtinbrengende bestanddelen bevatten.
- Filteren de luchtfilters van echografiesystemen COVID-19?
-
Net als andere medische apparaten zijn de koelsystemen en systeemfilters van Philips ontworpen om te zorgen voor een goede werking van de machines in diverse ziekenhuissituaties en klinische omgevingen. De filters van het koelsysteem zijn ontworpen om ophoping van stof en deeltjes in onze systemen te voorkomen, maar zijn niet ontworpen voor het filteren van pathogenen. De meeste systeemfilters van echografieproducten van Philips kunnen door de gebruiker worden verwijderd en kunnen worden gereinigd en gedesinfecteerd. We verwijzen u naar de officiële richtlijnen van CDC over de transportclassificatie en -mechanismen voor het SARS-CoV-2-virus.
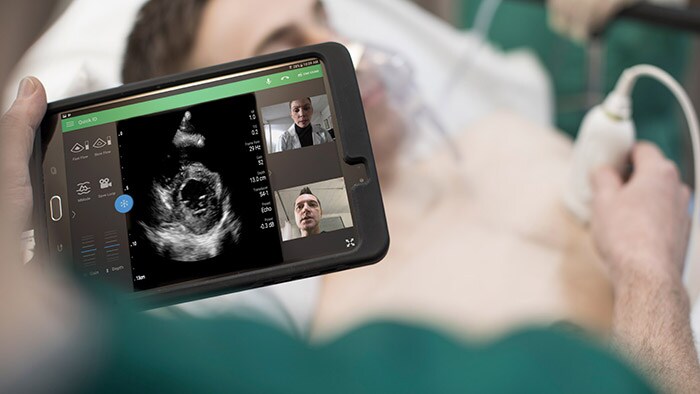
- Welke transducers beveelt Philips aan voor longechografie?
-
Er zijn variaties qua voorkeur. Voor kleine patiënten zijn Linear Array-transducers meestal het beste, maar voor de buik werkt Curved goed. Curved Array-transducers zijn het meest geschikt voor de longen; harttransducers werken echter goed en kunnen zowel de longen als het hart scannen.
- Heeft Philips de productie van beademingsapparaten en andere producten voor kritieke zorg verhoogd?
-
Philips werkt 24 uur per dag aan het verhogen van de productie van ziekenhuisbeademingsapparaten en andere producten voor kritieke zorg om patiënten met het nieuwe coronavirus (COVID-19) te helpen diagnosticeren en behandelen. Zoals aanvankelijk in maart is gecommuniceerd, zijn we onze productie van ziekenhuisventilatoren in mei aan het verdubbelen en zullen we tegen het derde kwartaal van 2020 een viervoudige toename hebben gerealiseerd.
Onze inspanningen om de productie te verhogen omvatten onder meer:
- Het inhuren van extra productiemedewerkers en het overplaatsen van huidige medewerkers om de toegenomen vraag onmiddellijk te ondersteunen;
- Het toevoegen van productielijnen en het verhogen van het aantal shifts naar 24/7 shifts;
- Nauwe samenwerking met leveranciers van Philips om de materiaaltoevoer te waarborgen die nodig is voor de toegenomen productie op onze productielocaties;
- Het benutten van de innovatiemogelijkheden van Philips om verwante productreeksen te herbestemmen om te voorzien in de toegenomen vraag;
- Samenwerking op contractbasis met de externe fabrikanten Jabil en Flex
Onze meest recente verklaringen vindt u in het Philips Nieuwscentrum.
- Welke andere producten en diensten biedt Philips voor de diagnose en behandeling van COVID-19?
-
Op dit moment is er het meest behoefte aan onze monitoren voor vitale functies en aan draagbare ventilatoren, en ook aan medische verbruiksartikelen voor niet-invasieve en invasieve beademing voor de behandeling van een breed scala aan luchtwegaandoeningen.
De producten en oplossingen van Philips voor de behandeling van COVID-19 omvatten onder meer:
- Point-of-care-oplossingen voor kritieke zorg ter behandeling van luchtwegaandoeningen. Dit zijn onder meer oplossingen voor bewaking van de vitale waarden van een patiënt en het detecteren van subtiele veranderingen, en ventilatoren en medische verbruiksartikelen voor invasieve, niet-invasieve en gecombineerde beademing voor de behandeling van een breed scala aan luchtwegaandoeningen.
- Diagnostische beeldvormingssystemen en -diensten, waaronder CT, mobiele diagnostische röntgenologie en echografie om luchtwegaandoeningen te helpen diagnosticeren.
- Telezorg-oplossingen voor ziekenhuizen voor het centraal bewaken en behandelen van patiënten op de intensive care (Philips eICU-programma) en telezorg-oplossingen voor het verbinden van zorgverleners en patiënten thuis.
Meer informatie over onze oplossingen voor de behandeling van COVID-19 vindt u hier.
- Hoe kan mijn ziekenhuis of overheid hulp krijgen van Philips?
-
Philips werkt actief samen met ziekenhuizen, de medisch-technologische industrie, overheden, de Wereldgezondheidsorganisatie en andere zorginstanties om de productie en verzending van levensreddende medische apparatuur en zorg veilig te stellen en te faciliteren, om tegemoet te komen aan de door COVID-19 veroorzaakte behoefte aan kritieke zorg. Als u nog geen klant bent of als u een overheidsvertegenwoordiger bent, kunt u hier contact met ons opnemen.
- Kan ik een beademingsapparaat van Philips kopen voor mezelf of een dierbare?
-
Invasieve en niet-invasieve beademingsapparaten van Philips zijn niet beschikbaar voor verkoop aan particulieren en kunnen alleen worden verkocht aan ziekenhuizen, leveranciers van medische apparatuur voor thuis (DME-bedrijven), instellingen voor langdurige zorg, medisch vervoer en overheidsorganisaties.
Philips biedt een breed portfolio van respiratoire en zuurstofproducten die verkrijgbaar zijn op voorschrift van een arts. U kunt ons portfolio van slaap- en respiratoire producten hier bekijken.
- Wat is de beste manier voor een ziekenhuis om contact op te nemen met Philips voor hulp of ondersteuning tijdens de COVID-19-pandemie?
-
Blijf alstublieft samenwerken met uw accountmanager of lokale verkoopvertegenwoordigers. Als u met een leidinggevende van Philips wilt spreken, kan uw accountmanager of lokale verkoopvertegenwoordiger u helpen deze gesprekken te faciliteren. Als u nog geen klant bent, neem dan hier contact met ons op.
- Welke hulpmiddelen voor training en technische ondersteuning zijn beschikbaar?
-
Voor aanvullende training in het gebruik van Philips-apparatuur hebben we tutorialvideo's ontwikkeld die u kunt openen via onze website over de COVID-19-respons van Philips.
Voor technische ondersteuning en productonderhoud raden wij u ten zeerste aan om Philips-beademingsapparaten alleen te laten onderhouden door getrainde onderhoudsmedewerkers die kennis van en ervaring met onze ventilatoren hebben en die de instructies in de servicehandleiding hebben gelezen en naleven.
- Wie kan onderdelen verkrijgen die nodig zijn om deze beademingsapparaten te repareren?
-
Klanten kunnen onderdelen rechtstreeks blijven bestellen via hun accountmanager of contactpersoon voor technische ondersteuning van Philips. Externe serviceproviders kunnen onderdelen blijven bestellen via distributeurs.
- Welke invloed zal COVID-19 hebben op de bedrijfscontinuïteit van Philips?
-
Philips heeft een Business Continuity Management System, dat is afgestemd op en gecertificeerd conform de internationale norm voor bedrijfscontinuïteit ISO 22301:2012. We zijn voortdurend bezig met de herziening van voorzorgsmaatregelen op basis van richtlijnen van de WHO en landelijke autoriteiten.
Ondanks de COVID-19-pandemie heeft Philips haar bedrijfsactiviteiten over de hele wereld kunnen voortzetten. Zoals verwacht, zien we een verminderde vraag naar de producten in ons consumentenportfolio in de meest getroffen gebieden en een grotere vraag naar ons professionele zorgportfolio.
Philips heeft een evenwichtige productie-impact over de hele wereld, met productielocaties in Noord-Amerika, Latijns-Amerika, Europa en Azië, waaronder diverse locaties in China. In overeenstemming met ons bedrijfscontinuïteitssysteem hebben we de relevante veiligheidsprotocollen geïmplementeerd op al onze wereldwijde locaties.
- Welke andere inspanningen doet Philips om door COVID-19 getroffen gemeenschappen te helpen?
-
De Philips Foundation werkt wereldwijd aan de strijd tegen de COVID-19-uitbraak in China, Kenia, Italië en daarbuiten. De Philips Foundation is het centrale platform voor de activiteiten van Philips op het gebied van maatschappelijk verantwoord ondernemen. De stichting is opgezet met de overtuiging dat we door innovatie en samenwerking een aantal van de moeilijkste uitdagingen in de wereld kunnen oplossen en impact kunnen hebben waar dat echt belangrijk is. Er verschijnen regelmatig updates in het Philips Nieuwscentrum.
Meest gezochte producten voor COVID-19-zorg
Tutorialvideo's voor COVID-19-behandeling
Philips Respironics V60 Gebruik voor invasieve beademing
Philips MobileDiagnost wDR Mobiel systeem voor digitale radiografie
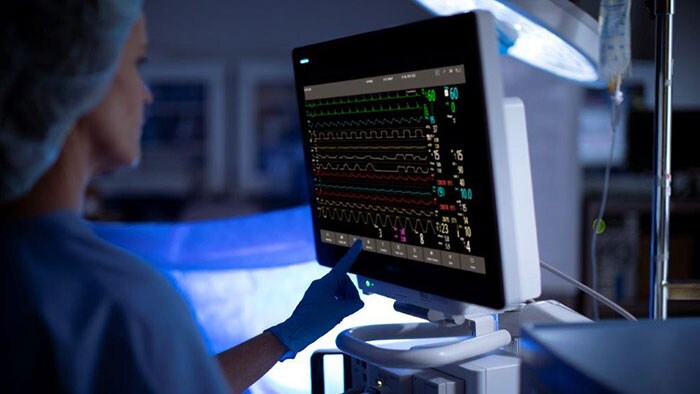
Philips IntelliVue-patiëntbewaking Basisfuncties
Philips IntelliVue-patiëntbewaking Informatie over alarmen
Probeert u ons te bereiken?

Ondersteuning medische producten Bel ons op 0800 0222350
Persvragen
Nieuws en visies
-
![Lessons from COVID-19: Improving care for cardiology patients]()
August 31, 2020
-
![How the COVID crisis could spark positive change in healthcare]()
August 11, 2020
Wij staan u bij

Georganiseerd om te helpen

Actief aanpassen
Verbindingen maken
Wij zien de inspanningen van de levensreddende beroepsgroepen om ons heen. Dit houdt ons hoopvol en motiveert ons om gefocust te blijven op onze kerntaken: het vervullen van essentiële klantbehoeften, het waarborgen van de gezondheid en veiligheid van werknemers, en het ondersteunen van het welzijn van mensen over de hele wereld.