apr 25, 2022
Philips maakt cijfers eerste kwartaal 2022 bekend
Philips realiseert in het eerste kwartaal een omzet van EUR 3,9 miljard, met 5% groei van de vergelijkbare orderontvangst als gevolg van de goede vraag
Kerncijfers eerste kwartaal Frans van Houten, CEO van Koninklijke Philips: “Onze klanten bevestigen de relevantie van onze strategie en ons portfolio, zoals blijkt uit de verdere groei van ons ‘all-time-high’ orderboek. De vergelijkbare groei van de orderontvangst voor de Groep bedroeg 5%, aangedreven door een goede groei in de Diagnosis & Treatment-activiteiten, evenals in Hospital Patient Monitoring en Connected Care Informatics. Daarnaast zijn we een partnerschap aangegaan met nog 12 ziekenhuizen om hen te helpen de zorgverlening te transformeren, voortbouwend op de 80 nieuwe strategische partnerschappen op lange termijn die in 2021 zijn aangegaan. In China hebben we een overeenkomst getekend met het Shanghai East Hospital om zijn ziekenhuizen in de Provincies Shandong en Hainan met een breed scala aan geavanceerde beeldvormings- en intensive care-oplossingen. Ik ben ook blij met de vergelijkbare omzetgroei van 8% voor onze Personal Health-activiteiten, wat een aanhoudend sterke vraag van consumenten naar onze proposities aantoont waarmee mensen voor hun gezondheid en welzijn kunnen zorgen.
Dankzij het harde werk van onze mensen boekten we een beter dan verwachte omzet van EUR 3,9 miljard in zeer uitdagende omstandigheden, met aanzienlijke tegenwind in de toeleveringsketen en de gevolgen van de veiligheidsmelding van Respironics. De aangepaste EBITA-marge voor de Groep bedroeg 6,2% in het kwartaal.
De sterke vraag van klanten en het sterke orderboek, gekoppeld aan onze verkoopprestaties in het eerste kwartaal, ondersteunen de groei en marge-uitbreiding voor het volledige jaar zoals gecommuniceerd in januari 2022. Tegelijkertijd is het belangrijk dat we de toenemende risico's erkennen die verband houden met de COVID-19 situatie in China, de oorlog tussen Rusland en Oekraïne, uitdagingen in de toeleveringsketen en inflatiedruk, die mogelijk van invloed kunnen zijn op ons vermogen om ons sterke orderboek om te zetten in verkopen en onze margedoelstelling te behalen als de omstandigheden verder verslechteren. Onze teams zijn volledig gefocust op de dagelijkse uitvoering, het voldoen aan de vraag van de klant en een sterk orderboek en het aanpakken van de supply chain-risico's. We voeren aanvullende kostenmaatregelen en prijsverhogingen door om de inflatoire tegenwind te verzachten.”
Business segment performance
The Diagnosis & Treatment businesses’ comparable sales decreased 2%, on the back of 9% comparable sales growth in Q1 2021. High- single-digit growth in Image-Guided Therapy was more than offset by a decline in Ultrasound and in Diagnostic Imaging due to electronic component shortages, and on the back of strong growth in these businesses last year. Comparable order intake increased 7%, with double-digit growth in Image-Guided Therapy and mid-single-digit growth in Ultrasound and Diagnostic Imaging, reflecting robust traction for Philips’ very attractive offering. The Adjusted EBITA margin was 5.9%, mainly due to the decline in sales and the impact of supply chain headwinds.
The Connected Care businesses’ comparable sales decreased 21%, mainly due to the consequences of the Respironics field action. Comparable order intake was in line with Q1 2021, with continued strong demand and share gains in Hospital Patient Monitoring and Connected Care Informatics. The Adjusted EBITA margin amounted to 0.4%, mainly due to the decline in sales and the impact of supply chain headwinds, partly offset by cost savings.
The Personal Health businesses’ comparable sales increased by a strong 8%, primarily driven by double-digit growth in Oral Healthcare. The Adjusted EBITA margin amounted to 15.3%, mainly due to the increase in sales, partly offset by supply chain headwinds and an adverse currency impact.
Philips’ ongoing focus on innovation and partnerships resulted in the following key developments in the quarter:
Cost savings
Our cost savings programs delivered EUR 97 million in the first quarter. After deducting supply cost increases, net savings amounted to EUR 8 million in the first quarter. In response to the inflationary headwinds, the company is implementing additional cost-saving measures of EUR 150-200 million for the full year.
Philips Respironics field action related to specific CPAP, BiPAP and mechanical ventilators
“We are committed to supporting the community of patients who rely on our sleep and respiratory care solutions for their health and quality of life, and the physicians and customers who are dedicated to meeting patient needs. We are replacing or repairing the devices related to the Respironics field action as fast as possible and are continuing to update patients and customers about the progress of the program. We have a strong program management in place overseeing every aspect of the remediation, which involves more than 1,000 of our colleagues,” said Frans van Houten, CEO of Royal Philips.
Philips has a strong program management in place led by Roy Jakobs, Chief Business Leader of the Connected Care businesses and member of Philips’ Executive Committee, to ensure the Respironics field action is executed with speed and accuracy. Management responsibility and oversight have been strengthened with organizational changes implemented in Philips Respironics and the Quality & Regulatory function. Staffing and expertise related to post-market surveillance, medical affairs, toxicology and bio-compatibility have also been increased.
Philips Respironics has more than tripled its weekly production output compared to 2020, despite the ongoing global supply chain challenges. To date, Philips Respironics has produced more than 2.2 million repair kits and replacement devices. Following another wave of Philips Respironics’ comprehensive patient and customer communication outreach and based on current insights, the total expected units to be remediated have increased by approximately 300,000, primarily in the US. Philips Respironics recorded a EUR 65 million increase in the field action provision in the quarter to cater for the higher expected volume of devices eligible for remediation and higher communication costs. Additionally, a further EUR 100 million provision was recorded for potential higher cost of execution and to ensure the speed of the program in a volatile environment. Philips Respironics expects to complete over 90% of the production and shipments to customers in 2022.
Philips Respironics continues to make good progress with the comprehensive test and research program to better characterize the possible health risks associated with the sound abatement foam in the affected devices. Comprehensive testing and analyses related to the affected CPAP and BiPAP devices are expected to be completed in the second quarter of 2022.
On April 8, 2022, Philips Respironics and certain of Philips’ subsidiaries in the US received a subpoena from the US Department of Justice to provide information related to events leading to the Respironics recall. The relevant subsidiaries are cooperating with the agency.
Report
Presentation
Conference call and audio webcast A conference call with Frans van Houten, CEO, and Abhijit Bhattacharya, CFO, to discuss the results will start at 10:00AM CET, April 25, 2022. A live audio webcast of the conference call will be available through the link below. Q1 2022 – First quarter 2022 results conference call audio webcast Click here for Mr. Bhattacharya's CV and images
More information about Frans van Houten and Abhijit Bhattacharya
Click here for Mr. van Houten's CV and images
Visit our interactive results hub for more on our financial and sustainability performance over the past quarter.
About Royal Philips
Royal Philips (NYSE: PHG, AEX: PHIA) is a leading health technology company focused on improving people's health and well-being, and enabling better outcomes across the health continuum – from healthy living and prevention, to diagnosis, treatment and home care. Philips leverages advanced technology and deep clinical and consumer insights to deliver integrated solutions. Headquartered in the Netherlands, the company is a leader in diagnostic imaging, image-guided therapy, patient monitoring and health informatics, as well as in consumer health and home care. Philips generated 2021 sales of EUR 17.2 billion and employs approximately 79,000 employees with sales and services in more than 100 countries. News about Philips can be found at www.philips.com/newscenter.
Forward-looking statements and other important information
Forward-looking statements
This document and the related oral presentation, including responses to questions following the presentation, contain certain forward-looking statements with respect to the financial condition, results of operations and business of Philips and certain of the plans and objectives of Philips with respect to these items. Examples of forward-looking statements include statements made about our strategy, estimates of sales growth, future Adjusted EBITA*), future restructuring and acquisition- related charges and other costs, future developments in Philips’ organic business and the completion of acquisitions and divestments. Forward-looking statements can be identified generally as those containing words such as “anticipates”, “assumes”, “believes”, “estimates”, “expects”, “should”, “will”, “will likely result”, “forecast”, “outlook”, “projects”, “may” or similar expressions. By their nature, these statements involve risk and uncertainty because they relate to future events and circumstances and there are many factors that could cause actual results and developments to differ materially from those expressed or implied by these statements.
These factors include but are not limited to: Philips’ ability to gain leadership in health informatics in response to developments in the health technology industry; Philips’ ability to transform its business model to health technology solutions and services; macroeconomic and geopolitical changes; integration of acquisitions and their delivery on business plans and value creation expectations; securing and maintaining Philips’ intellectual property rights, and unauthorized use of third-party intellectual property rights; Philips' ability to meet expectations with respect to ESG-related matters; failure of products and services to meet quality or security standards, adversely affecting patient safety and customer operations; breaches of cybersecurity; Philips' ability to execute and deliver on programs on business transformation and IT system changes and continuity; the effectiveness of our supply chain; attracting and retaining personnel; COVID-19 and other pandemics; challenges to drive operational excellence and speed in bringing innovations to market; compliance with regulations and standards including quality, product safety and (cyber) security; compliance with business conduct rules and regulations; treasury and financing risks; tax risks; reliability of internal controls, financial reporting and management process. For a discussion of factors that could cause future results to differ from such forward-looking statements, see also the Risk management chapter included in the Annual Report 2021.
Philips has recognized a provision related to the voluntary recall notification in the US/field safety notice outside the US for certain sleep and respiratory care products, based on Philips’ best estimate for the expected field actions. Future developments are subject to significant uncertainties, which require management to make estimates and assumptions about items such as quantities and the portion to be replaced or repaired. Actual outcomes in future periods may differ from these estimates and affect the company's results of operations, financial position and cash flows.
Third-party market share data
Statements regarding market share, contained in this document, including those regarding Philips’ competitive position, are based on outside sources such as specialized research institutes, industry and dealer panels in combination with management estimates. Where information is not yet available to Philips, market share statements may also be based on estimates and projections prepared by management and/or based on outside sources of information. Management's estimates of rankings are based on order intake or sales, depending on the business.
Market Abuse Regulation
This press release contains inside information within the meaning of Article 7(1) of the EU Market Abuse Regulation. This press release was distributed at 07:00 am CET on April 25, 2022.
Use of non-IFRS information
In presenting and discussing the Philips Group’s financial position, operating results and cash flows, management uses certain non-IFRS financial measures. These non-IFRS financial measures should not be viewed in isolation as alternatives to the equivalent IFRS measure and should be used in conjunction with the most directly comparable IFRS measures. Non-IFRS financial measures do not have standardized meaning under IFRS and therefore may not be comparable to similar measures presented by other issuers. A reconciliation of these non-IFRS measures to the most directly comparable IFRS measures is contained in this document. Further information on non-IFRS measures can be found in the Annual Report 2021.
Fair value information
In presenting the Philips Group’s financial position, fair values are used for the measurement of various items in accordance with the applicable accounting standards. These fair values are based on market prices, where available, and are obtained from sources that are deemed to be reliable. Readers are cautioned that these values are subject to changes over time and are only valid at the balance sheet date. When quoted prices or observable market data are not readily available, fair values are estimated using appropriate valuation models and unobservable inputs. Such fair value estimates require management to make significant assumptions with respect to future developments, which are inherently uncertain and may therefore deviate from actual developments. Critical assumptions used are disclosed in the Annual Report 2021. In certain cases independent valuations are obtained to support management’s determination of fair values.
Presentation
All amounts are in millions of euros unless otherwise stated. Due to rounding, amounts may not add up precisely to totals provided. All reported data is unaudited. Financial reporting is in accordance with the accounting policies as stated in the Annual Report 2021 except for the adoption of new standards and amendments to standards which are also expected to be reflected in the company's consolidated IFRS financial statements as at and for the year ending December 31, 2022.
Prior-period amounts have been reclassified to conform to the current-period presentation; this includes immaterial organizational changes.
*) Non-IFRS financial measure. Refer to Reconciliation of non-IFRS information.
Onderwerpen
Contact

Ben Zwirs
Philips Global Press Office Tel: +31 6 1521 3446
You are about to visit a Philips global content page
Continue
Derya Guzel
Philips Investor Relations Tel: +31 20 59 77055
You are about to visit a Philips global content page
ContinueBusiness Highlights Q1 2022
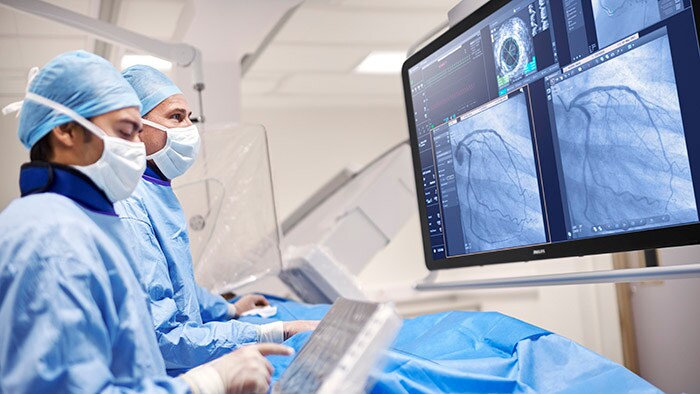
New long-term strategic partnerships
Philips signed 12 new long-term strategic partnerships in the quarter, including a 10-year agreement with Oulu University Hospital in Finland to deliver the latest Philips Azurion image-guided therapy solutions.
Expansion of capabilities on handheld ultrasound
Philips expanded its leading ultrasound portfolio with advanced hemodynamic measurement capabilities on its handheld ultrasound Lumify.

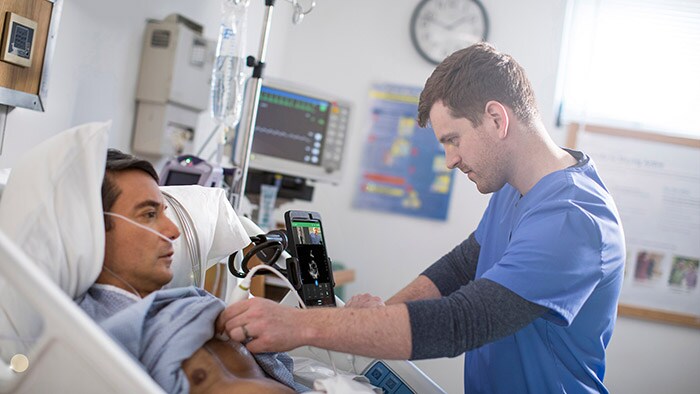
Research highlights benefits of remote cardiac monitoring
Underlining the clinical and economic value of remote cardiac patient monitoring, Philips announced new research demonstrating increased atrial fibrillation detection and significant cost savings using Philips’ mobile cardiac outpatient telemetry monitoring.

Completion of global roll-out of innovative shaver
Philips completed the global introduction of its new Philips Shaver S9000 with SkinIQ with its launch in Japan.